|
.
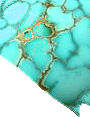
Limonite Mineral Facts:
Chemical Formula: Fe4O3(OH)6
Hydrated oxide of iron, often impure. The analyses
of limonite range between wide limits, largely because of the great
quantities of impurities mixed with it. The principal impurities are
clay, sand, phosphates, silica, manganese compounds and organic matter.
Once an important iron ore known in the mining trade "brown hematite" or
"bog iron ore".
Colors:
Various shades
of brown; when earthy, red, yellow or brownish-yellow.
Hardness:
5.0 to 5.5
Density: 3.6
to 4.0
Cleavage:
None.
Crystallography: Monoclinic
Limonite occurs in mammillated or stalactitic forms, having a radiating
fibrous structure, resembling that of hematite ; also in dull earthy
or massive condition and in concretions and psuedomorphs.
Luster:.
Submetallic to earthy, opaque.
|

Above: Limonite Pseudomorph after Pyrite |
|
.
Composition,
Structure and Associated Minerals:
Limonite is a common ore of iron and is always secondary in its origin,
formed through the alteration or solution of previously existing iron
minerals.
Pyrite
is often found altered
to limonite, the crystal form being at times preserved, giving limonite
pseudomorphs.
Sulfide
veins
are often capped near the surface, where oxidation has taken place, by a
mass of cellular limonite, which is known as gossan, or an iron hat. Iron
minerals existing in the rocks are among the first to undergo decomposition,
and their iron content is often dissolved by percolating waters through the
agency of the small amounts of carbonic acid which they contain.
The iron is transported as a carbonate by the waters to the surface and then
often carried by the streams finally into marshes and stagnant pools. There,
under the effect of the evaporation of the water and its consequent loss of
the carbonic acid, which served to keep the iron carbonate in solution, and
through the agency of the reducing action of carbonaceous matter present,
the iron carbonate is changed to an oxide, which separates from the water
and collects first as an iridescent scum on the surface of the water, and
then later
sinks to the bottom. In this way, under favorable conditions, beds of impure
limonite can be formed in the bottom of marshes and bogs.
|

Limonite |
|
|
Such
deposits are very common and are known as bog-iron ores, but, because of
the foreign materials deposited along with the limonite, are seldom of
sufficient purity to be worked. Limonite deposits are also to be found
in connection with iron bearing limestones. The iron content of the
limestone is gradually dissolved out by circulating waters and
transported by them to some favorable spot, and there the iron is slowly
redeposited as limonite, gradually replacing the calcium carbonate of
the rock. Or, by the gradual weathering and solution of the limestone,
its iron content may be left in the form of residual masses of limonite,
lying in clay above the limestone formation. Banded iron deposits
are often of considerable size, and because of their greater purity are
much more often mined than the bog-iron ores. Limonite is the coloring
material of yellow clays and soils, and mixed with fine clay makes what
is known as yellow ocher. Limonite is commonly associated in its
occurrence with
hematite, turgite,
pyrolusite,
calcite,
siderite, etc.
Identification and Diagnostics
In its chemical properties limonite resembles
goethite, from which it can be distinguished only with great
difficulty except when the latter is
in crystals. From uncrystallized varieties of goethite it can usually be
distinguished only by quantitative analysis, although in pure specimens
the streaks are different. Strongly magnetic after heating in reducing
flame. Yields much water in closed tube (15 per cent). Characterized
chiefly by its structure and yellow-brown streak.
Occurrence,
Localities and Origins:
Limonite is the
usual result of the decomposition of other iron-bearing minerals.
Consequently, it is often found as pseudomorphs.
The varieties recognized are: compact, the stalactitic and other fibrous
forms; ocherous, the brown or yellow earthy, impure variety; bog iron, the
porous variety found in marshes, pseudomorphing leaves, etc., and brown clay
ironstone, the compact, massive or nodular form.
In almost all cases where large
beds of the ore occur the material has been deposited from ferriferous water
rich in organic substances. One of the commonest types of occurrence is "gossan."
In the production of this type of ore, those portions of veins carrying
ferruginous minerals are oxidized under the influence of oxygen-bearing
waters, forming a layer composed largely of limonite which covers the upper
portion of the veins and hides the original vein matter. Gossan ores derived
from
chalcopyrite and pyrite are common in all regions in which these
minerals occur. Another type of limonitic iron ore comprises those found in
clays derived from limestones by weathering. In such deposits the ore occurs
as nodules and in pockets in the clay. Ores of
this type are common in the valleys within the Appalachian Mountains.
Bog iron ores occur in swamps and lakes into which ferruginous solutions
drain. At
times, bog iron ores are formed as beds on the floors of some lakes, as in
Sweden, where a layer of 7 inches thick accumulated in twenty-six years. The
deposition of the iron compound from the stream water flowing into the lake
may be caused by minute organisms. Some limonite beds are true chemical
precipitates.
The
iron may come from pyrite or iron silicates in the drainage basins of the
lakes or swamps. When carried down it is oxidized by the air and sinks to
the bottom.
Localities. The mineral occurs abundantly and in many different localities.
Deposits of the
bog iron type are to be found chiefly along the Appalachian Mountains, from
western Massachusetts as far south as Alabama. These ores have been of
considerable importance in western Massachusetts,
at Salisbury and Kent, Conn.; at many points in New Jersey,
southeastern
New York,
Pennsylvania, Michigan, Tennessee, Alabama, Ohio, Virginia and Georgia.
Limonite deposits of various kinds are found throughout the western part of
the US, but as yet they have not been extensively developed.
Although containing less iron than hematite, on account of its
cheapness, and the ease with which it works in the furnace, limonite was
once an important ore of this metal. The earthy varieties are used as
pigments in cheap paints.
Return to the
Mineral Collectors Information Page |
|